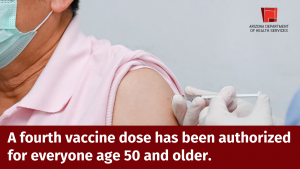
 Today, the Food and Drug Administration (FDA) and Centers for Disease Control and Prevention (CDC) authorized a second booster dose for those ages 50 and older. They also authorized a second booster for those 12 and older who have weakened immune systems.
Today, the Food and Drug Administration (FDA) and Centers for Disease Control and Prevention (CDC) authorized a second booster dose for those ages 50 and older. They also authorized a second booster for those 12 and older who have weakened immune systems.
Vaccines are extremely effective at protecting against severe COVID-19 outcomes, but evidence suggests the protection wanes over time. That’s why it’s so important to update your immune response with a COVID-19 booster dose.
For both groups authorized for a second booster, the recommendation applies at least four months after the first booster dose.
Boosters make a difference:
- During the Omicron surge, only a tiny fraction of cases, hospitalizations, and deaths in Arizona involved people who were fully vaccinated with a booster. In January alone, those who were fully vaccinated with a booster were 11 times less likely to test positive for COVID-19, 67 times less likely to be hospitalized with COVID-19, and 180 times less likely to die from COVID-19.
- Both groups authorized for a second booster dose have a greater chance of severe outcomes when breakthrough cases occur in vaccinated individuals. Despite this, nearly 40% of eligible Arizonans ages 65 and older have yet to receive their first booster dose.
If you are vaccinated but not yet boosted, ADHS encourages you to change that. Boosters are safe, highly effective and recommended at least five months after your Moderna or Pfizer series or at least two months after the Johnson & Johnson/Janssen shot.
If you are eligible for a second booster dose, please get one as soon as possible. The CDC notes that a second booster is especially important for ages 65 and older and those 50 and older with weakened immune systems. COVID-19 has been in decline lately, but getting boosted helps make sure your immune system is ready for whatever may come.
You’ll find hundreds of COVID-19 vaccine providers at azhealth.gov/FindVaccine. We answer many common questions about COVID-19 vaccines and boosters at azhealth.gov/COVID19Vaccines.